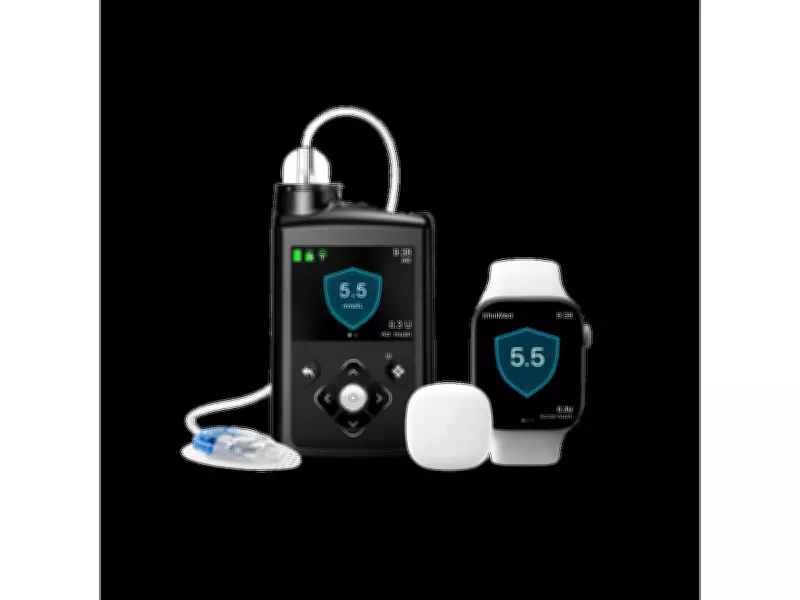
MiniMed Group, Inc. (Nasdaq: MMED), a global leader in insulin delivery, has announced that Health Canada has licensed new software for the MiniMed 780G system. This authorization represents a significant advancement in diabetes care for Canadians, offering greater flexibility through the integration of Meal Detection technology and SmartGuard with both the Guardian 4 and Simplera Sync sensors. Additionally, the system is now indicated for adults aged 18 and older with Type 2 diabetes and is compatible with Fiasp.
Simplera Sync Sensor Features
The Simplera Sync sensor is a disposable, all-in-one device that eliminates the need for fingersticks when used with SmartGuard. It requires no overtape and features a simple two-step insertion process, providing a more comfortable wear experience while reducing the daily burden of diabetes management.
How the MiniMed 780G System Works
The MiniMed 780G system employs an adaptive algorithm that automatically anticipates, adjusts, and corrects glucose levels every five minutes, operating around the clock. It is the only system with Meal Detection technology, which automatically detects rising glucose and adjusts insulin delivery to help maintain levels in range, even when users occasionally miss a dose or underestimate carbohydrate intake. The system uses a treat-to-target algorithm with flexible glucose targets as low as 5.5 mmol/L, closely mimicking the glucose levels of individuals without diabetes.
Clinical Evidence and Compatibility
Since its initial launch, the MiniMed 780G system has accumulated extensive clinical and real-world evidence across diverse populations worldwide. When using optimal settings—such as an active insulin time of two hours and a 5.5 mmol/L target glucose—the system has demonstrated the ability to help many people with Type 1 diabetes achieve over 80% time in range, exceeding the international target of 70%. It is also the only system compatible with the world's first seven-day infusion set, reducing changes to once weekly and delivering 96% fewer injections compared to multiple daily injections.
Industry Perspective
“This license is a milestone for the diabetes community in Canada,” said Chet Patel, Senior Business Director at MiniMed. “Our mission is to make every day a better day for people with diabetes. By continuing to evolve the MiniMed 780G system—including the addition of the Simplera Sync sensor and the indication for adults with Type 2 diabetes—we are closer to that vision than ever before.”
Availability
The Simplera Sync sensor will be available in Canada later this year. Currently, the MiniMed 780G system can be used with the Guardian 4 sensor.
About MiniMed
MiniMed is a global leader in insulin delivery, constantly advancing therapies that support people with diabetes in more than 80 countries. The company's full-stack, integrated ecosystem includes insulin delivery systems, continuous glucose monitors (CGMs), algorithms, and an easy-to-use app experience, all designed to work seamlessly together. With over 40 years of experience, MiniMed pioneers therapies that anticipate needs, reduce burden, and make life with diabetes easier.









