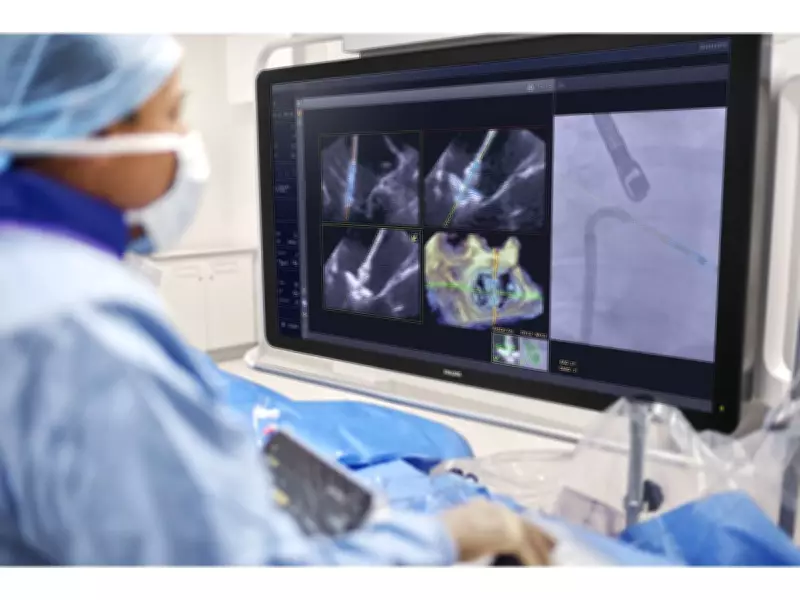
Royal Philips, a global leader in health technology, has achieved a significant milestone with the U.S. Food and Drug Administration (FDA) granting 510(k) clearance for its innovative AI-powered software solution. The newly approved EchoNavigator R5.0 with DeviceGuide is designed to assist physicians during one of interventional cardiology's most technically demanding procedures: repairing leaking mitral valves through a minimally invasive approach.
Revolutionizing Minimally Invasive Heart Valve Repair
DeviceGuide utilizes advanced artificial intelligence to track and visualize mitral valve repair devices in real time during complex procedures. This technology integrates seamlessly with the Philips Azurion image-guided therapy platform, which combines imaging, real-time data, and intelligent software within a unified workflow. The result is a system that supports more consistent, efficient, and confidently guided minimally invasive interventions, potentially transforming patient outcomes.
Collaborative Innovation with Edwards Lifesciences
The development of DeviceGuide was a collaborative effort between Philips and Edwards Lifesciences, the global leader in structural heart innovation. This partnership aligns Philips' expertise in imaging and AI with Edwards' deep knowledge in valvular heart therapy development. Together, the companies have innovated the image guidance of the mitral transcatheter edge-to-edge repair (M-TEER) workflow, making these intricate procedures more intuitive and streamlined for medical teams.
Addressing a Widespread Heart Condition
Mitral regurgitation (MR), or a leaking mitral valve, is a common heart condition affecting more than 35 million adults worldwide and over 2 million in the United States. Minimally invasive transcatheter techniques like M-TEER offer a valuable alternative to open heart surgery for select patient populations, reducing recovery times and surgical risks.
During M-TEER procedures, physicians make a small skin incision to access a vein, allowing a catheter to be introduced to the heart to deliver a repair device to the diseased mitral valve. The mitral valve's complex and heterogeneous structure requires experienced physicians to position the device delivery system accurately and guide the placement of the device with precision.
Enhancing Procedural Precision and Team Coordination
The intraprocedural heart team must interpret both X-ray and ultrasound images displayed on multiple screens, communicate effectively, and coordinate movements between two operators. They must make precise adjustments to grasp the moving valve leaflets and confirm results in real time, demanding exceptional accuracy, coordination, and experience from the entire team. DeviceGuide addresses these challenges by providing enhanced navigation guidance, potentially improving procedural outcomes and reducing complications.
Showcasing at a Premier Cardiovascular Forum
DeviceGuide will be prominently featured at the American College of Cardiology (ACC) 2026 meeting in New Orleans, one of the world's leading forums for cardiovascular innovation. This platform will allow medical professionals to explore the technology's capabilities and its potential impact on the field of interventional cardiology.
This advancement represents a significant step forward in medical technology, combining AI, imaging, and collaborative expertise to address a critical healthcare need. As minimally invasive procedures continue to evolve, tools like DeviceGuide may set new standards for precision and efficiency in heart valve repair.









