Alberta Health Services Explores Legal Action Over $49 Million Medication Contract Dispute
Alberta's provincial health authority is actively reviewing legal options to recover tens of millions of dollars from an outstanding contract with an Edmonton-based importer for Turkish-made pain medication, the majority of which has failed to materialize despite regulatory approvals and extensive negotiations.
Contractual Breakdown and Financial Implications
Alberta Health Services (AHS) confirmed this week that approximately $49 million remains outstanding under its agreement with MHCare, an Edmonton importer contracted to supply Turkish-manufactured medication. The health authority has abandoned previous attempts to fulfill the contract balance through alternative means and is now focusing on legal avenues to obtain reimbursement for the substantial financial commitment.
"AHS is reviewing legal remedies to obtain reimbursement," stated the agency in an official communication to media outlets. This represents a significant shift in strategy from earlier approaches that sought to salvage the agreement through modified import arrangements.
Regulatory Hurdles and Delivery Failures
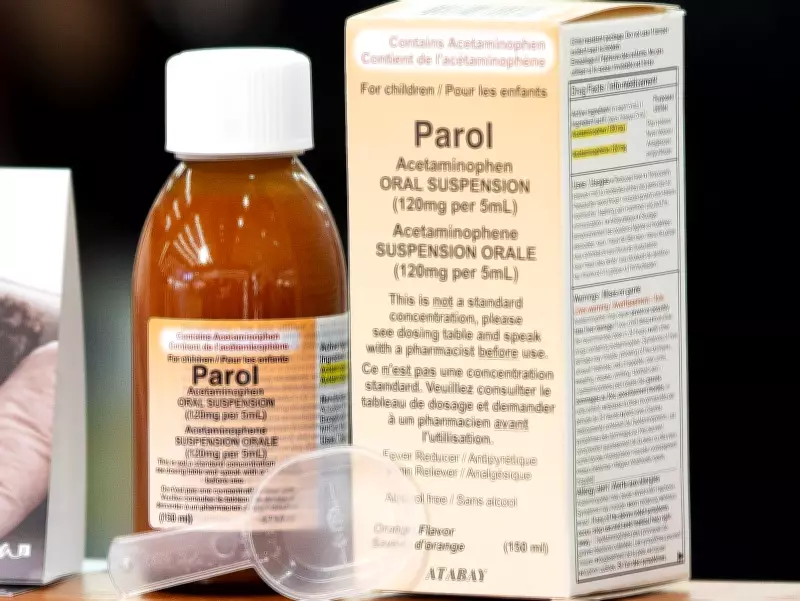
The contractual difficulties stem from Health Canada's approval process, which initially authorized the importation of 750,000 bottles of Parol (a Turkish pain medication) for behind-the-counter pharmacy sales across Alberta in early 2023. Despite this regulatory clearance, the actual delivery of medication has been minimal compared to the original agreement's scope.
Alberta had originally hoped to import five million bottles of the medication but only secured approval for 1.5 million units. Of these, just 11,000 bottles reached hospital facilities and 4,700 were delivered to pharmacies. The remaining inventory faced disposal as expiration dates approached in early 2026, with approximately 700,000 bottles donated internationally and over 800,000 destroyed.
Conflicting Perspectives on Contract Fulfillment
MHCare maintains a different position regarding the contract's status. Through legal representative Scott Hutchison, the company asserted: "MHCare remains of the view that it will be able to fulfill the terms of its contract with AHS in full, as has always been its intention." The importer attributes delays to Health Canada's regulatory review process, noting that a submission for acetaminophen review from manufacturer Atabay was accepted last August and remains under evaluation.
"Regulatory approvals can be frustratingly long but are beyond our control," Hutchison's statement continued, emphasizing that the contract terms were negotiated and approved by AHS at the highest organizational levels.
Political Acknowledgment and Administrative Response
Premier Danielle Smith acknowledged the changed approach during an unrelated news conference, stating: "As I understand that, they were looking at one pathway to get an alternate product, and it appears that they have exhausted that avenue, and AHS is now looking at means to be able to recover the outstanding balance." The Premier deferred further commentary to the health authority, which has declined additional statements beyond its initial position.
Historical Context and Previous Attempts at Resolution
This dispute follows months of attempted negotiations and alternative solutions. Last October, AHS indicated it was "assessing possibilities with the current agreement with MHCare" while emphasizing that "no decisions or purchases have been made, and we continue to review all options." The health authority had previously explored importing intravenous acetaminophen from the same Turkish manufacturer beginning in summer 2023, but has since abandoned that approach without reducing the outstanding financial obligation.
The situation highlights the complexities of international pharmaceutical procurement, regulatory compliance, and contract enforcement within Canada's healthcare system, with significant public funds at stake in the unresolved agreement.









