Canadian biotechnology company Eupraxia Pharmaceuticals Inc. has announced encouraging new long-term data from its ongoing clinical trial investigating a novel treatment for eosinophilic esophagitis (EoE), demonstrating consistent and durable symptom relief.
Long-Term Durability Confirmed
The latest 52-week follow-up data from Cohort 6 of the Phase 1b/2a RESOLVE trial aligns with previously reported long-term durability results from Cohort 5. These two cohorts represent the only patient groups that have reached the 52-week milestone in the study thus far.
According to the November 13, 2025 announcement, all three patients in Cohort 6 maintained clinical benefit from a single administration of EP-104GI, with two out of three patients remaining in clinical remission at the 52-week mark. When combined with data from Cohort 5, four out of six patients maintained clinical remission at one year post-treatment.
Consistent Symptom Improvement
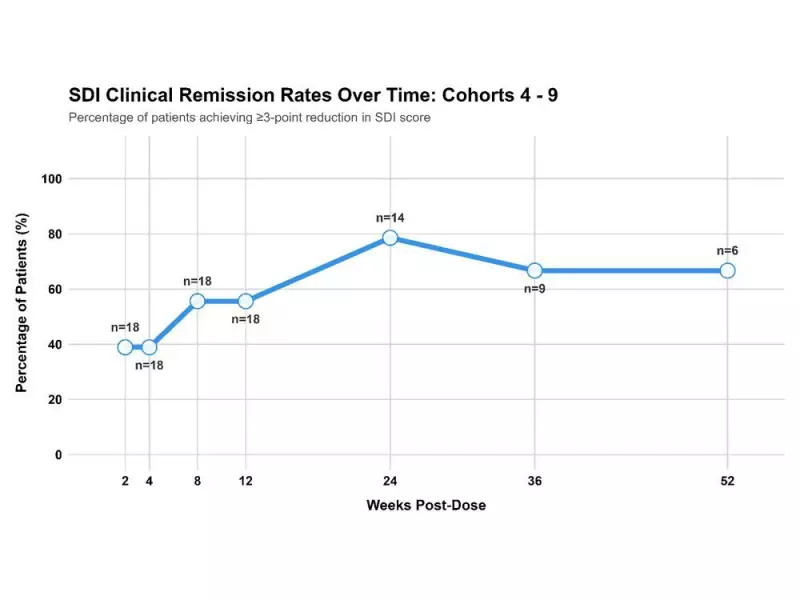
The trial measured symptom improvement using the Straumann Dysphagia Index (SDI), a standardized tool for assessing swallowing difficulties in EoE patients. In Cohort 6, which received a total dose of 64 mg EP-104GI, patients showed significant and sustained symptom relief.
Two of three patients in Cohort 6 achieved clinical remission at multiple time points, including week 12 (67%), week 24 (100%), week 36 (33%), and week 52 (67%). All patients maintained symptom improvements at week 52, with an average reduction in SDI scores of 3.7 points, representing a 58% improvement.
The broader patient population also showed strong results. At week 36, across Cohorts 5-7, 67% of patients were in clinical remission with an average SDI score reduction of 3.0 points (53% improvement). Even more impressive, at week 24 across Cohorts 4-8, 79% of patients achieved clinical remission with an average 3.7-point reduction (69% improvement) in SDI scores.
Promising Safety Profile
The treatment continues to demonstrate an excellent safety profile with now over 200 patient-months of follow-up across all cohorts. Notably, researchers reported no Serious Adverse Events and no cases of oral or gastrointestinal candidiasis, a common side effect of steroid treatments for EoE.
Pharmacokinetic data revealed that plasma levels of fluticasone in Cohort 6 patients remained constant and predictable throughout the 52-week period. These levels were well below those typically observed with daily asthma inhalers, though no head-to-head comparisons have been conducted.
Dr. James A. Helliwell, Chief Executive Officer of Eupraxia, emphasized the significance of these findings. "These data further highlight the strong durability and tolerability profile of EP-104GI, reinforcing its potential to become a convenient, once-a-year treatment that fits seamlessly into routine disease management by aligning with annual patient endoscopies," he stated.
Dr. Helliwell also noted the particular importance of a durable treatment option for EoE, explaining that "current EoE therapies often struggle with long-term adherence, we believe a durable, once-yearly treatment could meaningfully improve patient outcomes and establish EP-104GI as a preferred option for both physicians and their patients."
The company previously presented 52-week data as a late-breaking presentation at the American College of Gastroenterology Annual Scientific Meeting, with these new results building on that momentum. Eupraxia continues to advance its clinical program for EP-104GI, which leverages the company's proprietary Diffusphere™ technology designed to optimize local, controlled drug delivery.









