Prodeon Medical Achieves FDA Milestone with Urocross Expander System
Prodeon Medical, a privately held medical device company specializing in minimally invasive urologic therapies, has announced a significant regulatory achievement. The company has received U.S. Food and Drug Administration (FDA) 510(k) clearance for its innovative Urocross® Expander System, marking a new approach to treating lower urinary tract symptoms associated with benign prostatic hyperplasia.
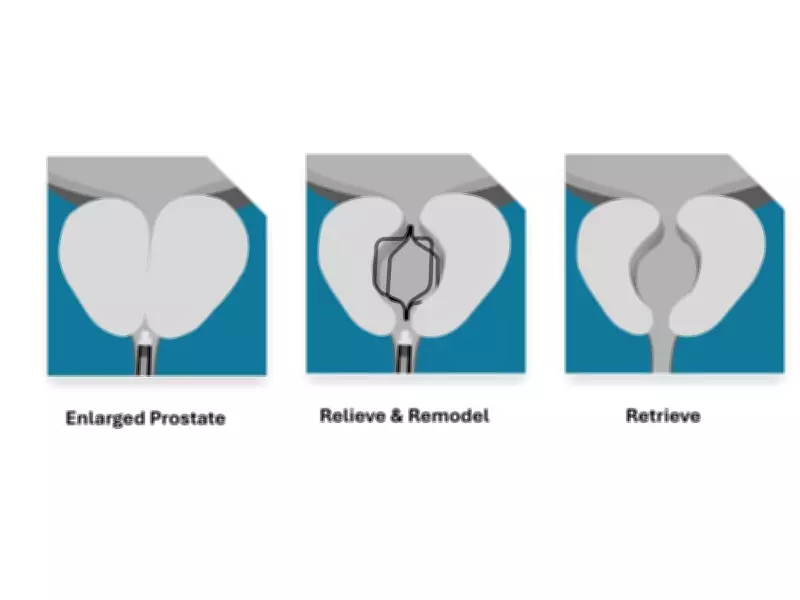
A Novel Approach to Prostate Treatment
The Urocross Expander System represents a departure from traditional BPH treatments by offering a non-permanent, retrievable implant solution. This technology is specifically designed to remodel obstructing prostatic lobes during a six-month indwell period before being intentionally retrieved, leaving no permanent foreign material in the patient's body.
The procedure utilizes commercially available flexible cystoscopes to place the temporary implant, which works to remodel the obstructed prostatic tissue. Unlike some existing treatments, the Urocross approach avoids cutting, burning, or ablating prostatic tissue, thereby preserving future diagnostic and therapeutic options for patients.
Clinical Data Supporting FDA Clearance
The FDA's decision was supported by comprehensive clinical data from multiple studies, including the Expander-1 Feasibility Trial and the Expander-2 Pivotal Trial. The latter was a large-scale, multi-center randomized controlled trial involving 240 patients across 23 sites in the United States and Canada.
Key findings from the clinical trials submitted for FDA clearance include:
- Mean improvement in International Prostate Symptom Score (IPSS) of 48.1% at 12 months post-implantation
- Procedural responder rate of 74.5%, defined as patients achieving ≥30% IPSS improvement at 12 months
- No device or procedural-related serious adverse events in the Urocross treatment arm
- Preservation of sexual function with no patients reporting sustained erectile or ejaculatory dysfunction
- High level of procedure tolerability comparable to diagnostic flexible cystoscopy
Long-Term Benefits and Clinical Implications
The available long-term clinical data demonstrates sustained improvements in urinary symptoms, quality of life, and urinary flow following Urocross retrieval. Importantly, these benefits were achieved without negative impacts on sexual function, addressing a common concern with many prostate treatments.
Dr. Kevin T. McVary, Professor of Urology at Loyola University Medical Center and Co-Principal Investigator of the Expander-2 Trial, emphasized the broader implications of this technology. "BPH is not solely a prostate condition; it is a bladder health issue," Dr. McVary stated. "Delaying intervention may contribute to irreversible bladder changes over time. New interventional technologies like Urocross enable minimally invasive approaches without permanent implants, providing safe and effective symptom relief with rapid recovery while preserving sexual function."
The Urocross System's temporary nature and preservation of tissue represent what Dr. McVary describes as "a meaningful evolution in BPH care" that might encourage earlier intervention by lowering the threshold for patients to seek treatment.
This FDA clearance positions Prodeon Medical's Urocross Expander System as a potentially transformative option in the management of benign prostatic hyperplasia, offering patients a minimally invasive alternative that balances effective symptom relief with preservation of future treatment options and sexual function.









