Helus Pharma, a clinical-stage pharmaceutical company focused on developing novel serotonergic agonists for serious mental health conditions, has announced promising topline results from its Phase 2 signal detection study evaluating HLP004 as a potential treatment for adults with moderate-to-severe generalized anxiety disorder.
Significant Clinical Improvement Demonstrated
The study revealed statistically significant and clinically meaningful improvement in anxiety symptoms, with patients receiving 20mg HLP004 adjunctive to standard of care therapy achieving a mean reduction of 10.4 points on the Hamilton Anxiety Rating Scale at six weeks. This improvement represents a substantial benefit above and beyond what patients were already experiencing with their existing treatment regimens.
Durable Effects and Strong Response Rates
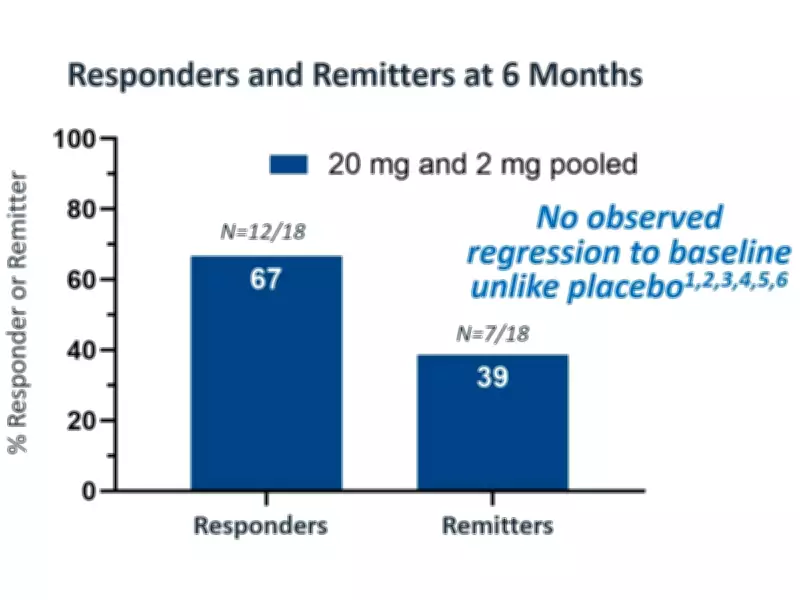
Perhaps most encouraging is the durability of these effects, which were sustained through at least six months of observation. At the six-month mark, the pooled study population demonstrated impressive response rates, with 67% of patients classified as responders and 39% achieving remission status.
The study specifically targeted a difficult-to-treat population consisting of moderate-to-severe patients who remained symptomatic despite ongoing antidepressant or anxiolytic therapy. This makes the positive results particularly noteworthy, as generalized anxiety disorder affects more than 20 million adults in the United States alone, with approximately half of treated patients failing to respond adequately to initial first-line therapy.
Practical Treatment Advantages
Beyond the clinical efficacy, HLP004 demonstrated several practical advantages that could make it commercially viable. The treatment features a short in-clinic experience with acute drug effects lasting approximately 90 minutes, and most participants were ready for discharge within three hours. This fits well within the treatment paradigm of existing interventional psychiatry clinics and represents a potentially scalable approach to anxiety treatment.
Favorable Safety Profile
The Phase 2 study also revealed a favorable tolerability profile for HLP004. Adverse events were generally transient, with no drug-related serious adverse events recorded and no suicidality-related safety signals detected. This safety profile is particularly important for anxiety treatments, where medication tolerability often influences patient adherence and treatment success.
The study involved 36 patients randomized in a 2-to-1 active-to-placebo ratio to receive either HLP004 20mg or 2mg doses administered via intramuscular injection three weeks apart. All participants continued their standard of care medications throughout the trial, with follow-up extending through Week 12 and observational follow-up continuing up to one year.
Addressing Unmet Medical Needs
These results are especially significant given the current treatment landscape for generalized anxiety disorder. No adjunctive pharmacologic treatment for GAD has ever received regulatory approval, and no new monotherapy has been approved in nearly two decades. The demonstrated efficacy of HLP004 as an adjunctive therapy therefore represents a potential breakthrough in anxiety treatment.
Participants in the study had an average baseline HAM-A score of 22 and a General Anxiety Disorder-7 score of greater than or equal to 10 at screening, indicating moderate-to-severe anxiety symptoms. The consistent improvement across multiple measures suggests HLP004 could offer meaningful relief for patients who have not responded adequately to existing treatments.









