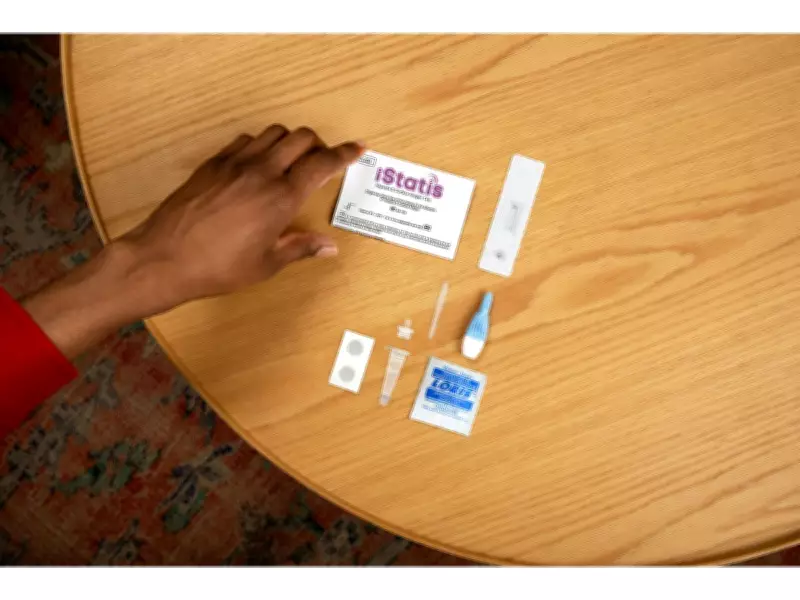
Health Canada has granted authorization to bioLytical Laboratories Inc. for the iStatis Hepatitis B Surface Antigen (HBsAg) Test, marking the first rapid point-of-care test for hepatitis B approved in North America. This innovative diagnostic tool is designed to deliver results in just 15 minutes, enabling timely detection and improved patient care in clinical and community settings.
A Critical Step in Hepatitis B Diagnosis and Prevention in Canada
Hepatitis B remains a significant yet underdiagnosed public health issue in Canada, with over 260,000 Canadians living with chronic hepatitis B. Alarmingly, nearly half of those infected are unaware of their status, according to public health estimates. The iStatis HBsAg Test addresses this gap by providing a portable, accurate, and easy-to-use solution for decentralized testing.
Addressing Gaps in Hepatitis B Diagnosis
Limited access to timely testing has been a persistent barrier, particularly in remote, underserved, and high-risk communities. Traditional lab testing often requires multiple visits and delays in results. With the iStatis test, healthcare providers can screen patients and deliver results during a single visit, facilitating quicker linkage to care and treatment.
“Too many people with hepatitis B remain undiagnosed because testing isn’t always accessible,” said Robert Mackie, CEO of bioLytical Laboratories. “This authorization allows screening to move closer to the patient, whether that’s in a clinic, a mobile unit, or a community outreach setting.”
A Tool for Frontline and Community-Based Care
The iStatis HBsAg Test detects the hepatitis B surface antigen, a marker of active infection, with high sensitivity and specificity. Designed for decentralized use, it requires minimal training and no additional instrumentation, making it suitable for a wide range of care environments, including harm reduction services and community health programs.
“Rapid, reliable screening at the point of care can make a meaningful difference in how quickly patients are diagnosed and linked to treatment,” said Dan Wang, Director of Scientific Affairs at bioLytical. “This test brings accurate hepatitis B screening directly to frontline healthcare providers.”
The authorization supports Canada’s public health goals and aligns with the World Health Organization’s target to eliminate viral hepatitis as a public health threat by 2030. By expanding access to rapid testing, the iStatis test is poised to play a crucial role in reducing the burden of hepatitis B across the country.